All published articles of this journal are available on ScienceDirect.
Panic Disorder and Chronic Caffeine Use: A Case-control Study
Abstract
Background:
Acute administration of caffeine produces panic attacks in most Panic Disorder (PD) patients, but little is known about chronic caffeine use in these patients.
Objective:
To assess caffeine use in patients with PD and to ascertain if caffeine consumption is associated with sociodemographic or clinical features.
Methods:
65 adults with PD and 66 healthy controls were included in the current study. Their caffeine intake within the previous week was quantified with a questionnaire and compared. Harmful caffeine use was defined as consumption above 400 mg/day of caffeine. We tested for correlations between caffeine intake, demographic and clinical features.
Results:
Patients consumed significantly more caffeine than controls (P < 0.001). 14% (N = 9) of the PD patients made harmful use of caffeine. The use of caffeine-containing medications was observed in 40% (N = 26) of the PD patients and 6% (N = 4) of controls. Consumption of energy drinks was observed in 11% (N = 7) of PD patients and in none of the healthy subjects. Patients reported sleeping significantly less than controls (P < 0.001). In PD patients, caffeine consumption was not correlated with the presence of panic attacks or comorbidity with depression. The use of benzodiazepines or sedative medications was not correlated with caffeine intake.
Conclusion:
High caffeine consumption in PD patients could be explained by the development of tolerance with regular use of this substance. Subtypes of sensitive and non-sensitive PD patients could also explain why some of these patients are able to tolerate high doses of caffeine.
1. INTRODUCTION
Panic Disorder (PD) is an anxiety disorder characterized by recurrent panic attacks (PA), fear of causes and consequences of these attacks, and avoidance [1]. Patients with this disorder are sensitive to anxiogenic/panicogenic affects of many substances, including lactate, carbon dioxide, cholecystokinin, yohimbine, and even antidepressants [2-4]. Several studies demonstrated that single doses of caffeine may induce anxiety and PA in these patients [2, 5, 6]. One would expect that PD patients would decrease or discontinue caffeine use due to its anxiogenic effects [7], however, many PD patients continue consuming this substance, what could be explained by habit, culture or an attempt to offset sedative effects of medications. In those with major depressive disorder comorbidity, caffeine could be used as self-medication due to its activating effects [7, 8].
Caffeine is one of the most consumed psychoactive substances in the world. Its effects include increased alertness and arousal, physical resistance, improvement of mood, tolerance to sleep deprivation and to pain, sociability, and well-being [7]. It is consumed in foods and beverages, such as chocolate, cocoa, soft drinks, coffee, tea, yerba mate and energy drinks. Caffeine may be included in analgesic formulations [9] and in some food supplements that supposedly increase energetic expenditure [10]. Population-based surveys in the US indicate that 85-95% of adults consume caffeine, and the mean intake is 193 mg/day [11], while in Brazil, considering only caffeine consumed in coffee, the mean intake is 238 mg/day [12].
Daily caffeine consumption above 400 mg could be associated with general toxicity, cardiovascular effects, behavioral changes, and other medical problems [13]. Acute use of caffeine may cause tachycardia, hypertension, irritability, anxiety, insomnia, and other symptoms [14]. A single dose of caffeine (400 to 480 mg) may induce PA in 35% to 48% of PD patients but does not induce PA in healthy subjects [5, 6]. By reducing sleeping hours, caffeine could indirectly cause headache, irritability, fatigue, and inability to concentrate [9]. Caffeine-induced sleep deprivation may also increase anxiety and PA [15]. In the study from Roy-Byrne et al. [15], one night's sleep deprivation induced PA in 40% of PD patients.
Adenosine is an inhibitory neurotransmitter present in the central nervous system and adenosine receptors, particularly A1 and A2Areceptors, are likely to be the major targets for caffeine [7]. Caffeine binds to these receptors and antagonizes the effects of adenosine. The activation of A1 receptors by adenosine can cause inhibition of adenylyl cyclase and voltage-sensitive Ca++-channels such as the N- and the Q-channels, and activation of several types of K+-channels, phospholipase C and phospholipase D. Consequently, a host of different cellular effects can ensue. Adenosine A1 receptors are present in almost all brain areas, with the highest levels in hippocampus, cerebral and cerebellar cortex, and certain thalamic nuclei. Only moderate levels are found in caudate-putamen and nucleus accumbens. The presence of presynaptic adenosine A1 receptors mediating inhibition of transmitter release has been demonstrated in virtually all types of neurons. Still, the mechanisms through which caffeine induces anxiety are not fully understood. It is known that adenosine, acting via A1 receptors, can regulate the release of many different neurotransmitters, including glutamate. If the effect of adenosine is blocked, excitatory transmission would be enhanced, which could directly or indirectly influence GABAergic transmission. Studies also demonstrated that high concentrations of caffeine can decrease the binding of benzodiazepines [7].
Apparently, susceptibility to caffeine-related adverse effects may be affected by the capacity to metabolize the substance, which is lower in individuals carrying the CYP1A2 polymorphism in the gene that codifies the cytochrome P450 oxidase [16]. The adenosine A2A receptor gene (ADORA2A) variation may also increase susceptibility to caffeine’s negative effects, such as anxiety and PA [17, 18]. In patients with this polymorphism, caffeine binds with high affinity to the adenosine A2A brain receptor [17], inducing PA after the consumption of caffeine doses as low as 150 mg [18]. In addition, Hohoff et al. [19] found that the variant rs5751876 of the ADORA2A gene is infact a risk factor for PD, particularly PD with agoraphobia. High family history of PD, which implies high genetic loading, was consistently observed in PD respiratory subtype patients [20]. Nardi et al. [21] found that these patients had more caffeine-induced panic attacks (odds ratio = 8.9; 95% confidence interval = 3.3 - 24.0) compared to non-respiratory subtype PD patients.
Currently, data on caffeine consumption and caffeine abuse in PD patients is still lacking. The primary objective of this study was to assess the consumption of caffeine in a clinical sample of PD patients and compare it with caffeine consumption in healthy subjects. The secondary objective was to ascertain if caffeine consumption is associated with sociodemographic or clinical features.
2. MATERIALS AND METHODS
This observational case-control study was conducted from July 2015 to July 2016 at the outpatient clinic of the Laboratory of Panic and Respiration at the Institute of Psychiatry of Federal University of Rio de Janeiro. Adults were eligible if they had at least two panic attacks in the month preceding inclusion. Healthy controls, matched for age and sex, were selected among staff and students and has no history of psychiatry disorders neither previous lifetime anxiety episode.Patients were excluded if they had bipolar disorder, schizophrenia or severe personality disorder, while controls were excluded if they used any sedative, antidepressant or antipsychotic medication within the week preceding the interview. Both patients and controls were excluded if they used any illicit drug within the week preceding the interview.
A detailed questionnaire on caffeine consumption and sleeping habits within the week before the interview was used in the assessment. The caffeine content of food and beverages was quantified according to the caffeine chart from Mayo Clinic [22]. The caffeine content of medications was obtained in the medications’ labels. Caffeine contents in analgesic formulations varied from 30 to 50 mg per dose. In thermogenic supplements and other food supplements the caffeine contents varied from 100 to 400 mg per dose.
According to the study from Nawrot et al. [13] the consumption of caffeine up to 400 mg/day does not present significant health risks, but above this level it could be associated with general toxicity, cardiovascular effects, changes in adult behaviour, increased incidence of cancer, effects on calcium balance or male fertility. In the current study the authors considered caffeine use above 400 mg/day as “harmful caffeine use”.
The study was approved by the institutional ethics committee. All subjects signed an informed consent form before inclusion in the study.
Continuous variables were compared between groups with Student's t Test, and categorical variables, with the Chi-Square Test. Spearman’s and point biserial correlations were used to check for correlations between variables. The level of statistical significance was set at 5%. IBM SPSS Statistics version 20 was used for data analysis.
3. RESULTS
The baseline characteristics of the 65 patients and 66 controls included in the study are shown in Table 1. Among PD patients, 68% had comorbidity with depression. Fifteen patients (23%) were not using antidepressants, benzodiazepines or other psychotropic medications. Of the 43 (65%) patients using antidepressants, 28 (42%) were being treated with selective serotonin reuptake inhibitors, 7 (11%) with serotonin and noradrenaline reuptake inhibitors, 8 (12%) with tricyclics, and 2 (3%) with other antidepressants. Forty-three patients (65%) were on benzodiazepines on a regular basis, 35 (53%) were using clonazepam, while 8 (12%) were using other benzodiazepines. A total of 35 (53%) patients were using both antidepressants and benzodiazepines. There were also patients on quetiapine, zolpidem, and topiramate, one patient using each of these drugs. None of the patients or controls were using melatonin or other sedatives.
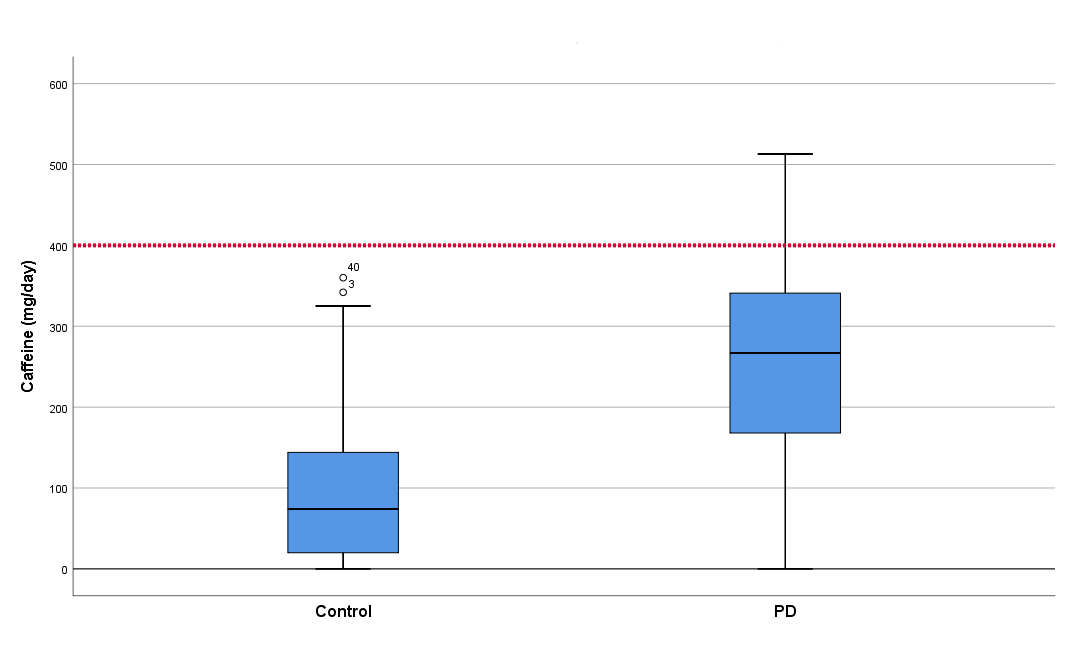
Daily consumption of coffee was reported by 41 patients (63%) and 46 controls (70%). Patients consumed significantly more caffeine than controls. PD patients reported using more caffeine-containing medications and energy drinks than controls. Nine patients (14%) and none of the controls were taking six or more pills of caffeine-containing medications per day, seven days a week. One patient (2%) was using thermogenic supplement pills (with high caffeine dose) before exercising, as an attempt to increase energy expenditure. There were nine (14%) PD patients and no controls with harmful caffeine use pattern. (Table 1) (Fig. 1).
Forty-seven percent of the PD patients had at least one PA in the week preceding the study. The caffeine consumption was 272 (SD = 131) mg/day for PD patients who had recent PA and 247 (SD = 121) mg/day for patients who did not have recent PA, there were no statistically significant differences between the two groups (Point biserial correlation 0.134, P = 0.287) (Fig. 2).
Comorbidity with depression, benzodiazepine use or use of sedative medications (amitriptyline, imipramine, nortriptyline, trazodone, quetiapine or zolpidem) were not correlated to caffeine intake in PD patients. In the whole sample analysis, caffeine intake was not correlated with gender or age.
Patients reported sleeping significantly fewer hours per night than controls and there was a strong negative correlation between the caffeine consumption and hours of sleep (Spearman’s rho: -0.478; P< 0.001) in the whole sample analysis. In the analysis of the PD group, caffeine intake was not correlated to hours of sleep.
There were reports from some patients that they tried at least once to quit caffeine abruptly because of sleep deprivation, headache or both, but their attempts resulted in the severe headache that remitted with caffeine-containing analgesics, or caffeine-containing food or beverages.
4. DISCUSSION
In the current study, we demonstrated that PD patients consume significantly more caffeine than healthy subjects. Interestingly, higher caffeine intake was not associated with panic attacks. This evidence is not in accordance with studies of single-dose administration of caffeine [5, 6], which demonstrated provocation of PA in a significant portion of PD patients. The difference found between acute and chronic use could be attributed to the establishment of tolerance to caffeine. One hypothesis would be that chronic caffeine users develop tolerance to this substance, and for this reason would not have PA even with high caffeine doses. Those who do not use caffeine regularly would remain sensitive to this substance. Another hypothesis, in agreement with the one from Nawrotet et al., [13] would be that PD patients are able to adjust their caffeine intake according to their individual sensitivity to this substance. One subgroup of patients would be sensitive to caffeine, and for this reason consume little or no caffeine. The other subgroup would not be sensitive to caffeine’s panicogenic effects, and would be able to consume large amounts of this substance. Genetic factors [18] or respiratory subtype [20] could be mediators to caffeine sensitivity in PD.
It is still not clear why PD patients would consume more caffeine than control subjects. Depression symptoms, which could include low energy, low motivation and hypersomnia, could prompt PD patients to use stimulating substances, including caffeine [23]. However, we did not find an association between major depressive disorder diagnosis and caffeine consumption. We also investigated the hypothesis that PD patients used caffeine as an attempt to counteract the sedative effects of benzodiazepines and other medications and found that caffeine consumption was not correlated to these medications.
| - | Patients | Controls | Statistic (P) | ||
|---|---|---|---|---|---|
| - | Mean/N | SD/% | Mean/N | SD/% | |
| Age | 41 | 12 | 40 | 14 | ns |
| Females | 43 | 66 | 44 | 67 | ns |
| Current smoker | 7 | 11 | 2 | 3 | ns |
| Daily caffeine intake (mg) | 259 | 126 | 100 | 90 | <0.001 |
| Use of caffeine-containing medications | 26 | 40 | 4 | 6 | <0.001 |
| Consumption of energy drinks | 7 | 11 | 0 | - | 0.007 |
| Harmful caffeine use | 9 | 14 | 0 | - | <0.002 |
| Hours of sleep | 5.1 | 2.0 | 7.5 | 1.3 | <0.001 |

An Italian study [24] explored and compared caffeine intake in patients with psychiatric disorders and healthy subjects and in terms of current caffeine consumption, they did not differ, but the lifetime intake of caffeine was significantly higher in the patient group. It is important to highlight that this sample includes patients with major depressive disorder, bipolar disorder (type I and II), panic disorder, obsessive-compulsive disorder, social anxiety disorder, schizophrenia, bulimia, anorexia, and binge eating disorder. Perhaps this configuration of the sample explains this difference.
Some studies speculate that neuroesteroid 3alpha, 5alpha-THP through positive modulation of GABA receptor activity may produce a regulatory mechanism against caffeine-induced anxiety, so neuroactive steroids may modulate the stimulant and anxiogenic effects of caffeine [25, 26].
As demonstrated in the current study, PD patients consume considerable amounts of caffeinated products, and some of them consumed more than 400 mg per day, which is the maximum recommended dose. Making recommendations and providing information about the negative effects of caffeine is important for our patients to make decisions. Regular caffeine use superior to 500-600 mg/day could result in “caffeinism”, which is a syndrome characterized by several adverse reactions including restlessness, anxiety, irritability, agitation, muscle tremor, insomnia, headache, diuresis, sensory disturbances, cardiovascular and gastrointestinal symptoms [13]. Even lower doses could produce health problems, such as decreased fertility in males and obstetric complications during pregnancy [13].
An important limitation of this study is that data on caffeine consumption, sleeping habits, and panic attacks were collected by self-reported questionnaire, which is subject to recall bias. A more precise way to assess caffeine consumption would be the measurement of its serum level, but that was not done in the current study. The researchers did not use specific clinical scales to assess depression symptoms or PD symptoms. This study could be underpowered to identify correlations with low effect sizes in the PD group analysis. Since this is a cross-sectional study, we were able to check correlations, but it was not possible to identify the causes and the effects. Other limitation is the fact that in our study, we did not access the lifetime caffeine consumption of both groups neither the duration of illness of patients group.
CONCLUSION
Caffeine consumption in PD patients was high compared to healthy controls. However, high caffeine intake was not correlated to panic attacks, depression, sedative medication use or sleeping hours. Future research should elucidate if regular caffeine use decreases the sensitivity to caffeine’s panicogenic effects or if there are subgroups of PD patients with low and high sensitivity to caffeine. In a clinical setting, it is important to inform patients about the problems that could arise with regular consumption of large doses of caffeine, even if caffeine does not interfere with anxiety and panic symptoms.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the ethics committee of Institute of Psychiatry of Federal University of Rio de Janeiro, Brazil.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Written and informed consent was obtained from all the participants prior to the study.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
The first author, Veruska Santos received a scholarship from the Brazilian Council for Scientific and Technological Development (CNPq).
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


